Pathogens invoke immune responses and can cause disease or even death to the host. According to the recent statistics from the World Health Organization, pathogens claim tens of millions of lives annually. To monitor, predict, and limit the impact of pathogens, rapid quantification of the pathogen concentration in different matrices is of great importance.

Toward this aim, droplet microfluidics-based droplet digital loop-mediated isothermal amplification (ddLAMP) was developed to detect and quantify pathogens by isothermally amplifying a nucleic acid sequence that is specific to the pathogen. The ddLAMP exhibits a high specificity, a fast amplification speed and can tolerate the presence of inhibitors, making ddLAMP suitable for the pathogen quantification. However, performing ddLAMP requires costly external flow control systems and adequate experience in droplet microfluidics, impeding more widespread adoption of this assay, particularly in resource-limited laboratories.
To overcome this limitation, we developed a ddLAMP assay, featuring significantly decreased operational difficulty and instrumentation requirements. The proposed assay consists of a hand-powered microfluidics design, which we call “Handifluidics” and can emulsify the LAMP mixture into highly monodisperse droplets just by manually pulling a connected syringe, without the need for costly flow control systems. After emulsification, the droplets are directly collected and then heated to conduct the LAMP reaction and the copy number of the target nucleic acid can be determined by imaging these droplets under a fluorescence microscope and calculating the fraction with positive signals. Throughout the whole process, neither droplet microfluidic expertise nor precise pressure or flow control systems is required. Moreover, multiple Handifluidics devices can be operated simultaneously to perform multi-sample detection, which is difficult to achieve in current ddLAMP techniques.
To verify the performance of the Handifluidics design, we mannually pull the syringe connected to the Handifluidics, and find the generated droplets to be highly monodisperse, with a droplet size coefficient of variation (CV) of 1.7%, comparable to the CV achieved using traditional droplet microfluidics or droplet microfluidic-like devices, which rely on costly flow control systems. In addition, the detection limit can be decreased to as low as 10 copies/µL. Therefore, we believe the proposed ddLAMP assay can increase the accessibility of this technique to more laboratories, particularly those that are resource-limited.
This work has been published and featured on the Supplementary Cover on ACS sensors. The team would like to acknowledge and appreciate the financial support from the Ministry of Science and Technology (MOST) and NTU’s Higher Education Sprout Project.
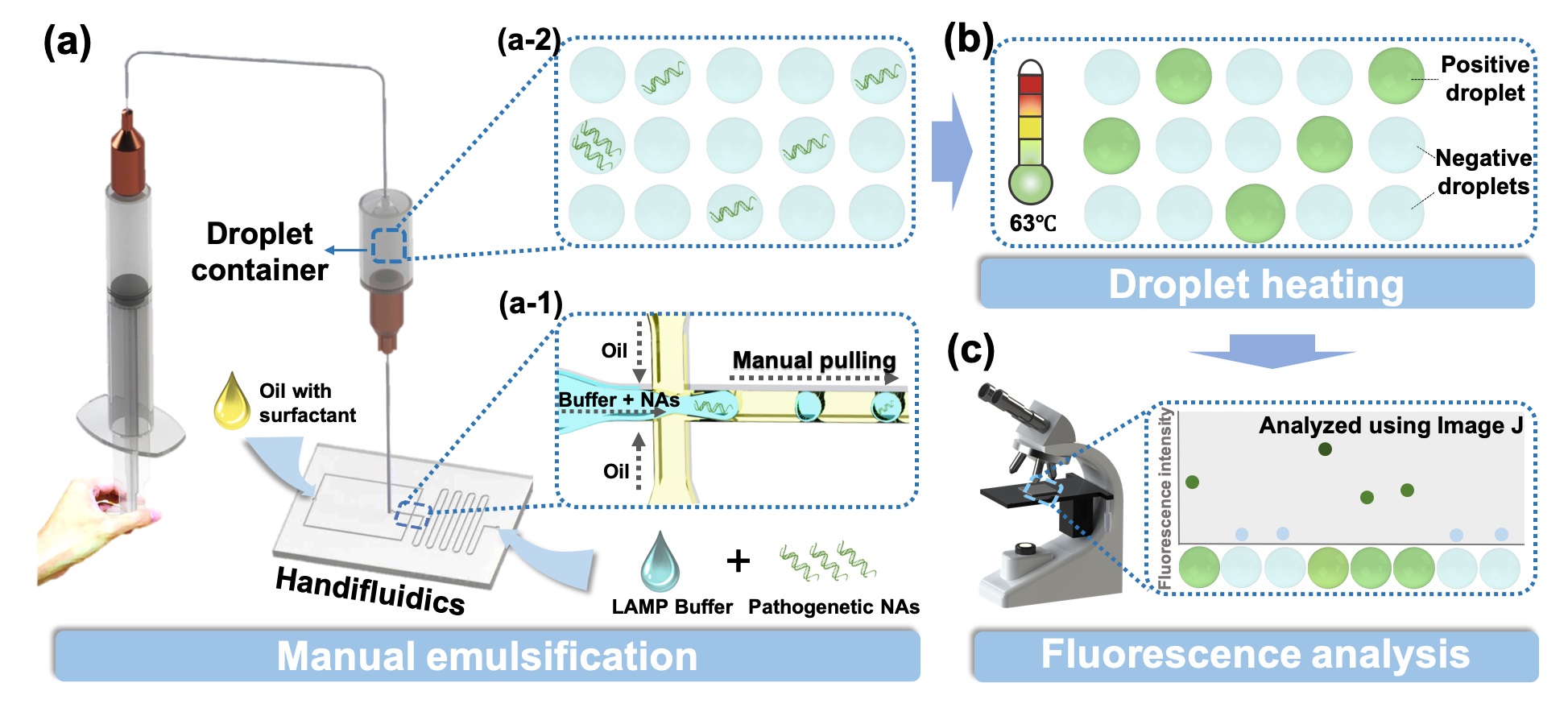
Schematic of the operating procedure of the proposed ddLAMP assay. (a) Schematic of the manual emulsification process. The LAMP mixture is emulsified into monodisperse droplets (a-1) and collected into a droplet container (a-2) by manually drawing a syringe to generate a negative pressure that drives the Handifluidics system. (b) Schematic of the droplet heating process. (c) Schematic of the droplet fluorescence analysis.
Contact: Prof. Chien-Fu Chen
This email address is being protected from spambots. You need JavaScript enabled to view it.
More information:
“Hand-Powered Microfluidics for Parallel Droplet Digital Loop-mediated Isothermal Amplification (ddLAMP) Assays,” ACS sensors
